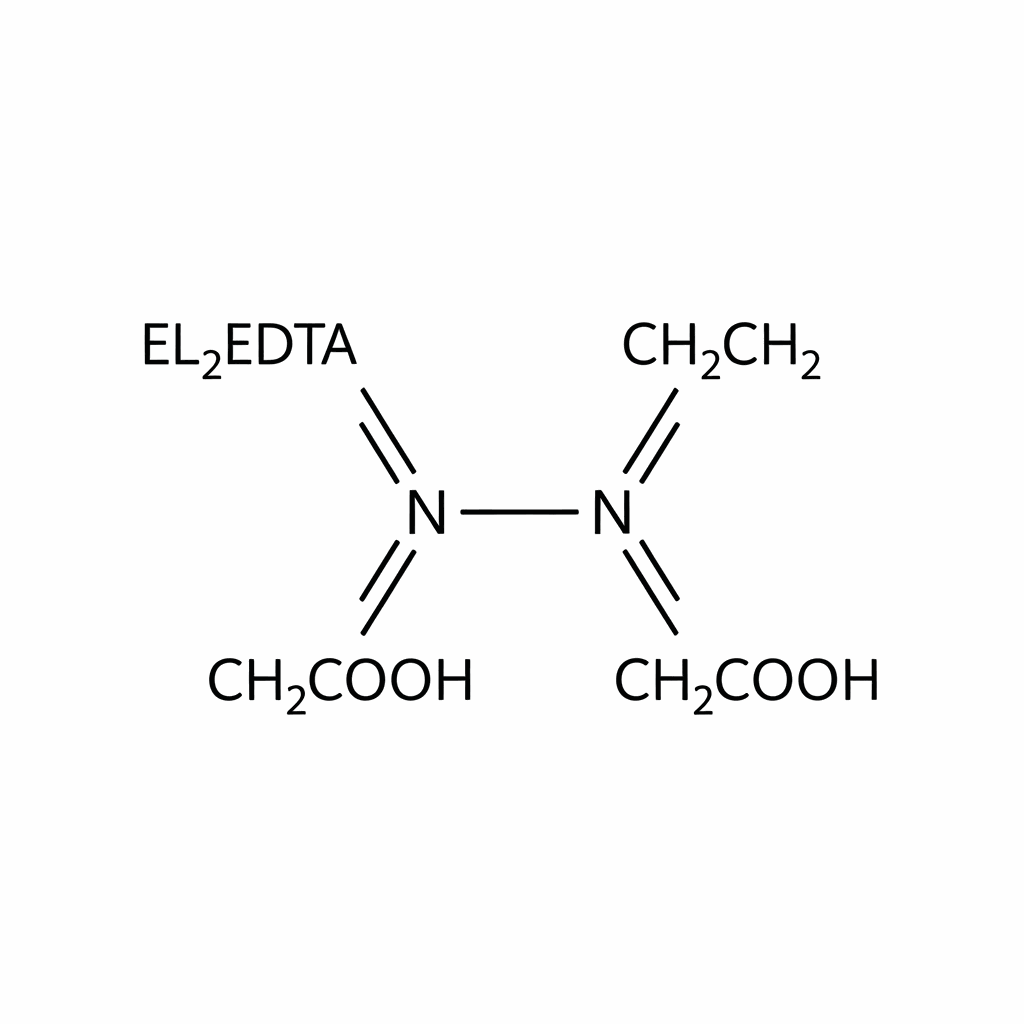
Ethylenediaminetetraacetic acid, commonly known as EDTA (Acid Form, CAS: 60-00-4), is a fundamental aminopolycarboxylic acid and one of the most versatile hexadentate chelating agents in modern chemistry. It appears as a white, odorless, crystalline powder. The primary chemical utility of EDTA lies in its unique ability to “sequester” or bind to polyvalent metal ions—such as calcium ($Ca^{2+}$), magnesium ($Mg^{2+}$), and iron ($Fe^{3+}$)—forming stable, water-soluble complex rings. This process effectively prevents these metal ions from participating in undesirable chemical reactions, such as oxidation catalysis or the formation of insoluble precipitates (scale). While the pure acid form (CAS 60-00-4) has limited solubility in water compared to its sodium salts (such as Disodium EDTA, CAS 6381-92-6), it is preferred in applications requiring high purity or precise pH adjustments. It serves as a critical stabilizer and decontaminant across industrial, pharmaceutical, and analytical sectors.




Have a question or need a quote? Our team is here to help with product details, pricing, and global delivery options.
Your email address will not be published. Required fields are marked *