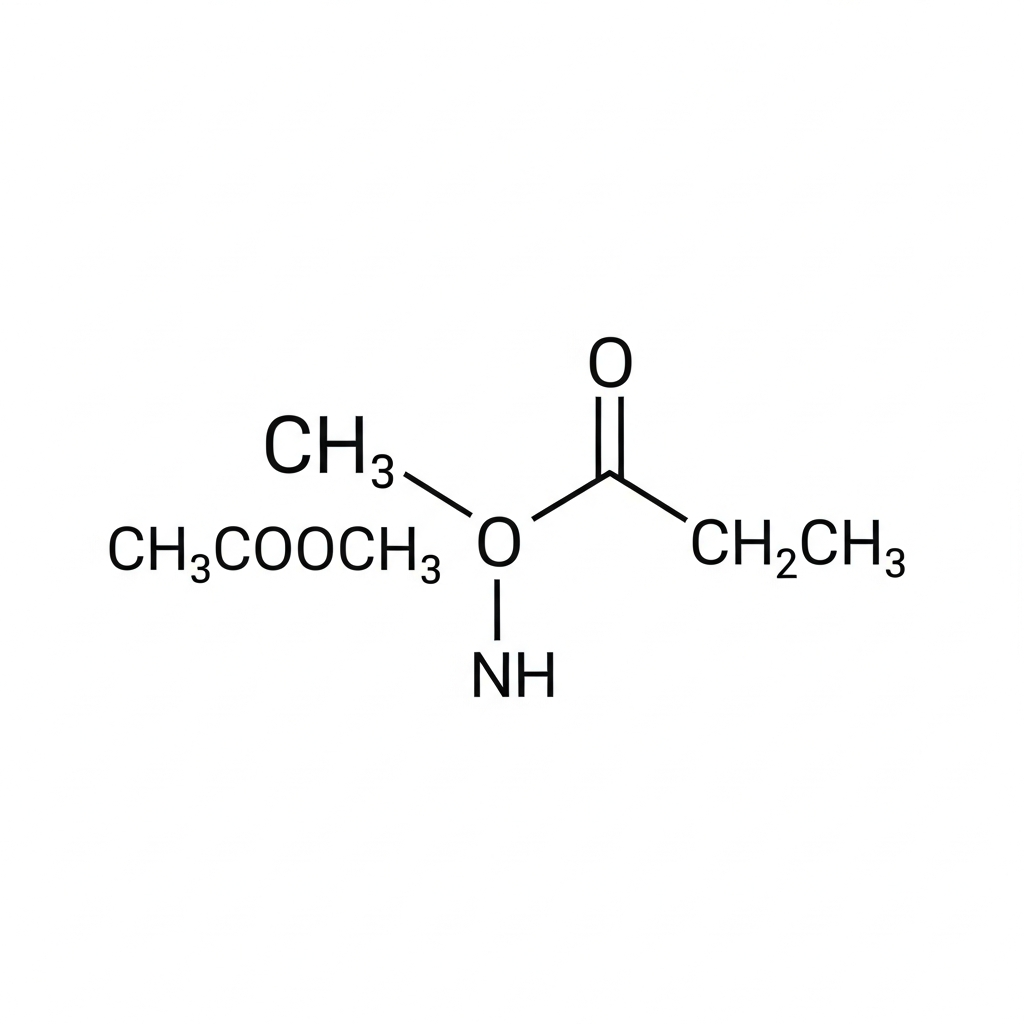
Ethyl acetate (CAS: 141-78-6), also known as ethyl ethanoate, is a significant organic ester widely utilized for its versatile solvent properties and characteristic fruity aroma. Chemically represented by the formula CH3COOCH2CH3, it is synthesized via the Fischer esterification of ethanol and acetic acid, typically in the presence of an acid catalyst. As a polar aprotic solvent, it provides an optimal balance between high solvency power and low toxicity, often serving as a safer alternative to aromatic or chlorinated hydrocarbons. Its relatively low boiling point and high volatility facilitate rapid evaporation, which is a critical requirement in industrial coating and printing processes. Beyond synthetic applications, ethyl acetate occurs naturally in various fruits and fermentation byproducts. It is valued in laboratory settings for liquid-liquid extraction and chromatography. While classified as a highly flammable liquid, its high biodegradability and low bioaccumulation potential make it an environmentally preferred choice in modern chemical manufacturing.




Have a question or need a quote? Our team is here to help with product details, pricing, and global delivery options.
Your email address will not be published. Required fields are marked *