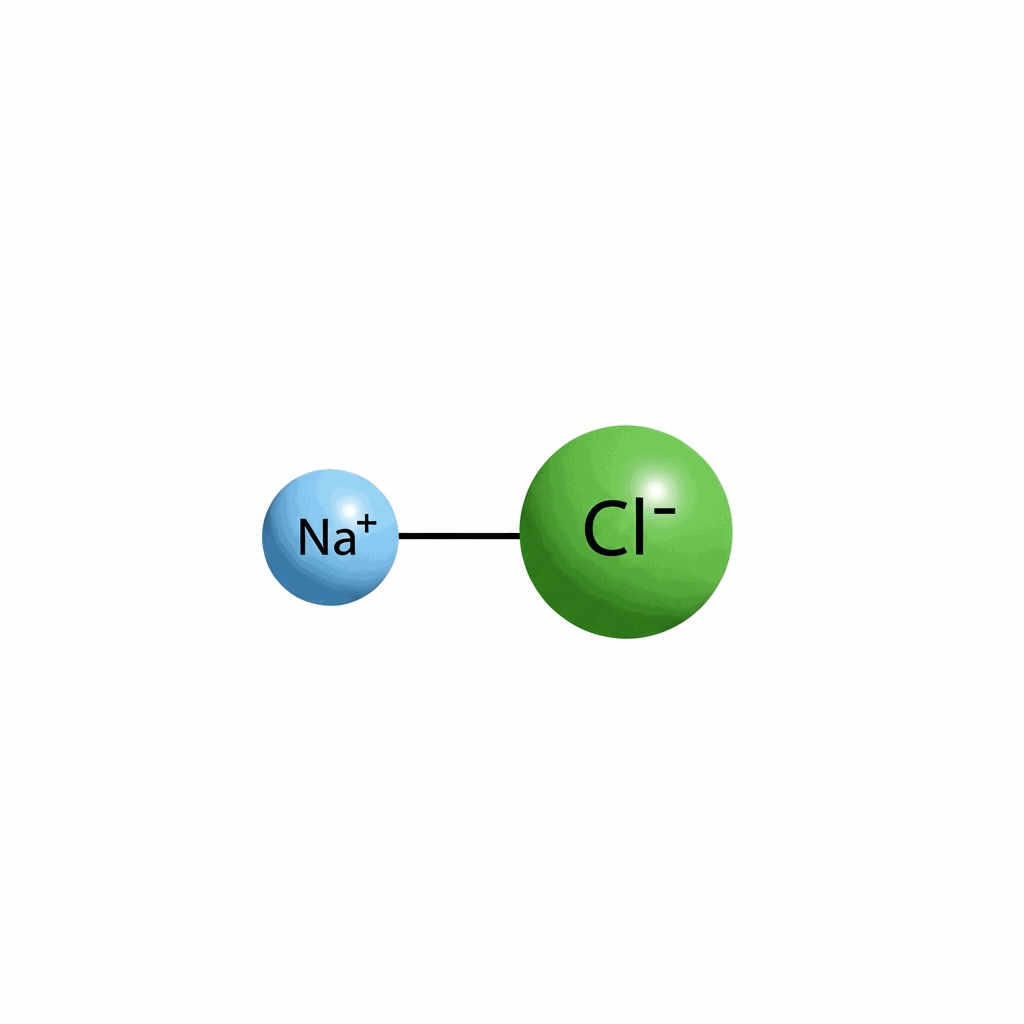
Sodium chloride, commonly recognized as table salt or halite, is an essential ionic compound with the chemical formula NaCl, representing a 1:1 stoichiometric ratio of sodium and chloride ions. This white crystalline solid is ubiquitous in nature, found in vast underground deposits and dissolved in high concentrations within seawater. As a fundamental electrolyte, it plays a critical role in biological processes, maintaining osmotic pressure and fluid balance in living organisms. In industrial sectors, sodium chloride serves as a primary raw material for the production of essential chemicals, including chlorine, sodium hydroxide, and sodium carbonate through processes like electrolysis. Its physical properties, such as high water solubility and thermal stability, make it indispensable across diverse fields ranging from food preservation to metallurgy. Historically, it has been a driver of global trade and economic development. Today, sodium chloride remains a cornerstone of modern chemistry, supporting infrastructure through de-icing and enhancing the industrial efficiency.




Have a question or need a quote? Our team is here to help with product details, pricing, and global delivery options.
Your email address will not be published. Required fields are marked *