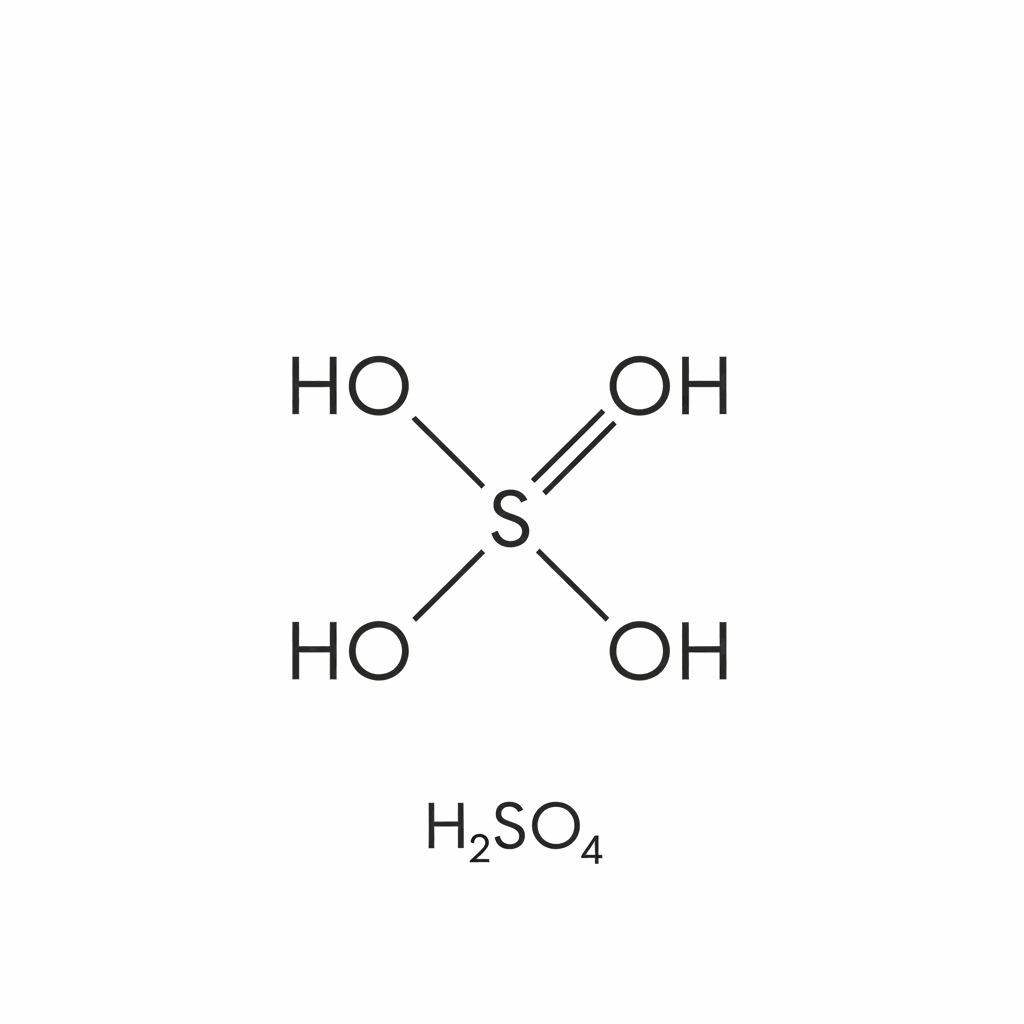
Sulfuric acid (H2SO4) is a highly corrosive, dense, oily liquid that is one of the most commercially important chemicals globally. Known historically as oil of vitriol, it is a strong mineral acid with powerful dehydrating and oxidizing properties. Its production volume often serves as an indicator of a nation’s industrial strength. The acid is colorless to slightly yellow, depending on purity, and is fully miscible with water in an extremely exothermic reaction. Primarily produced via the contact process involving the oxidation of sulfur dioxide, it serves as a fundamental building block in chemical synthesis. Its molecular structure consists of a central sulfur atom bonded to two hydroxyl groups and two oxygen atoms. Because of its high affinity for water, it is frequently used as a drying agent. Handling requires extreme caution due to its ability to cause severe chemical burns and permanent tissue damage upon contact, necessitating rigorous safety protocols.




Have a question or need a quote? Our team is here to help with product details, pricing, and global delivery options.
Your email address will not be published. Required fields are marked *